Many of the dental materials and devices handled by Shofu are classified as "medical devices" under the Act on Securing Quality, Efficacy and Safety of Products Including Pharmaceuticals and Medical Devices in Japan, as well as under applicable regulations in other countries. Because these products are primarily used in the oral cavity and must meet strict safety and biocompatibility requirements, a high level of quality control and quality assurance is essential.
As a company that manufactures and handles medical devices, Shofu views it as its mission to provide a stable supply of safe, reliable, and high-quality products. In line with this mission, the Company has established the following Quality Policy.
Quality Policy
To contribute to dental care around the world, Shofu complies with relevant regulatory requirements and provides a stable supply of high-quality products with a strong sense of ethics.
To achieve this, we set quality objectives, operate our quality management system appropriately, and continually improve all aspects of quality.
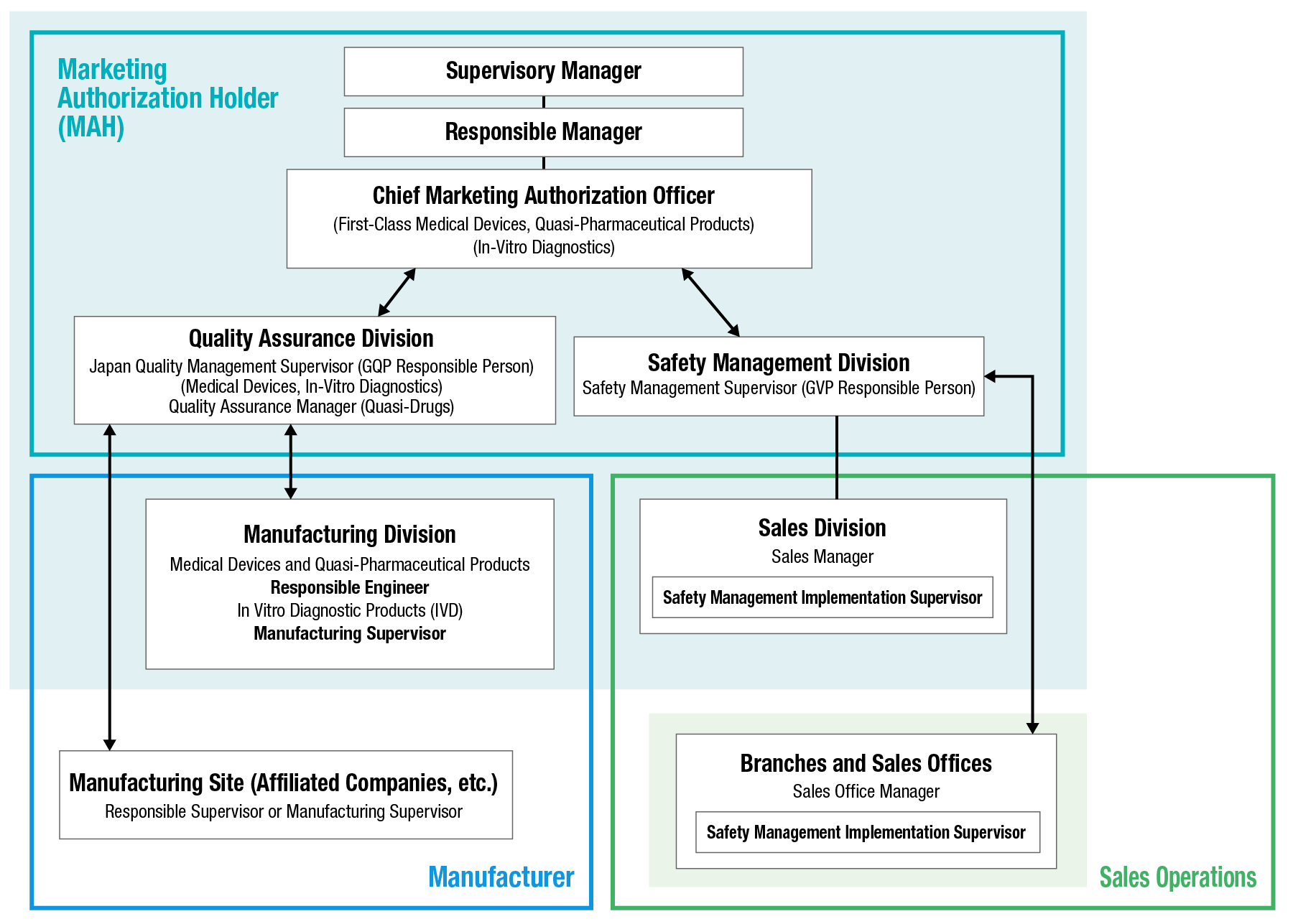
Quality Management System Aligned with Global Medical Device Regulations
In 1997, Shofu became the first company in the Japanese dental industry to obtain CE marking certification by meeting the requirements of the Medical Device Directive (MDD: 93/42/EEC), a quality standard established by the European Union (EU). We have also built a quality management system that conforms to the high standards of ISO 9001, the international standard for quality management systems, and ISO 13485, which specifies additional requirements for medical devices.
To ensure a stable supply of high-quality and high-value-added products and further enhance reliability, we continuously improve our quality assurance framework and post-market safety management system. In addition, to complying with The Pharmaceutical and Medical Device Act (The Act on Securing Quality, Efficacy and Safety of Products Including Pharmaceuticals and Medical Devices) in Japan, we meet standards and regulatory requirements established in countries around the world to deliver safe and reliable dental materials and equipment globally.
Quality Assurance Activities Supporting Product Safety and Reliability
Dental materials must be tailored to the needs of individual patients, making high-mix, multi-product manufacturing a fundamental requirement. Accordingly, it is essential to ensure reliable quality for every product we deliver.
To implement consistent quality assurance activities from research and development through post-market stages, we set quality objectives for each business process--including design and development, procurement, manufacturing, and customer-related processes--and carry out continuous improvement initiatives across the entire Company.
To ensure that our quality management system operates continuously, appropriately, and effectively, we conduct an annual management review in which senior management evaluates quality-related performance. The results of this review are reflected in the following year's activities, enabling us to maintain and strengthen quality assurance practices that support the safety and reliability of our products.
Safety Management System
To ensure that dental professionals can use our products with confidence, Shofu collects safety-related information--such as reports of malfunctions--from both domestic and overseas sources in accordance with the GVP Ordinance. We evaluate and review this information to assess its impact on the quality, effectiveness, and safety of our medical devices. Information about quality, effectiveness, and safety obtained through these processes is promptly and appropriately communicated to dental professionals to promote the proper use of our products. Through these activities, we strive to minimize safety-related risks and to continuously enhance the safety and reliability of our products.
- GVP Ordinance: Standards for post-marketing safety management of drugs, quasi-drugs, cosmetics, medical devices, and regenerative medical products.
Employee Training
Shofu provides training on its quality management system to enhance employees' awareness of quality and deepen their understanding of quality-related practices. In addition, we offer training necessary for maintaining and improving our quality management system, including GMP/QMS courses and safety management information programs for employees in specific roles. Training related to the QMS is conducted not only through external seminars offered by specialized institutions but also through in-house sessions led by external instructors and internal seminars conducted by employees with advanced expertise in quality management.
- GMP: Standards for manufacturing and quality control of pharmaceuticals
- QMS: Standards for manufacturing and quality control of medical devices and in vitro diagnostic products
Production System Capable of Handling Small-Lot, Multi-Product Manufacturing
Dental equipment must be tailored to the needs of each individual patient, resulting in a wide variety of products handled by the Company. To flexibly manufacture a diverse range of high-quality products, it is essential not only to introduce state-of-the-art production equipment, but also to establish proprietary manufacturing technologies for each product and to enhance the skills of the employees involved in production.
At Shofu, while promoting automation of production equipment and systems, we place strong emphasis on improving operator skills and ensuring smooth technology transfer through detailed instruction and a well-structured support system. We have also established a production flow in which only conforming products proceed to subsequent processes, and we continually work to eliminate bottlenecks and inefficiencies throughout the entire workflow--from order receipt to manufacturing and delivery.



